
ABF-STEM image of BaTiO3
News & Events

CIME News
Inauguration of a new high-end Transmission Electron Microscope at the EPFL-Valais campus on May 22nd.

Seminars
View all upcoming and past seminars
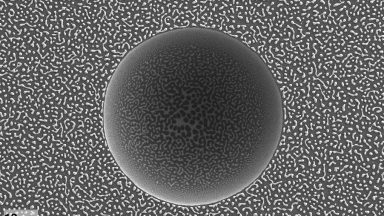
Image contest
The winner of January/February contest period is: “Lunar eclipse” by Laurène Tribolet (FIMAP). Congratulations!
Contact
Feel free to contact us by email and we’ll get back to you as soon as possible.
Email: [email protected]
Secretariat: +41 21 693 44 05
Address: EPFL MXC-130, Station 12, CH-1015, Lausanne





